Oligometastatic Disease in Real-World Radiology: The ECR 2026 Award-Winning Study That Reveals a Critical Gap
Reading time /
4 min
Company News

A large-scale analysis of 33.7 million radiology reports uncovers how rarely radiologists independently document oligometastatic disease and why that matters for patient outcomes.
At ECR 2026, the European Congress of Radiology in Vienna, the world's second-largest radiology congress, one of Segmed’s studies stood out. Among 11,376 submitted abstracts, an observational study through real-world data analysis of oligometastatic disease (OMD) documentation in routine U.S. radiology practice was recognized with the Best Research Presentation Abstract Award, on the topic of Oncologic Imaging. For a field still debating whether AI or structured reporting will close the gap between imaging interpretation and clinical decision-making, this study reframes the question entirely. The gap is not technological: it is linguistic, habitual, and measurable. And it is happening right now, in routine practice, across millions of reports.
This blog breaks down what the study found, why it matters, and what it signals for the future of structured reporting and AI-assisted oncologic imaging.

The Researcher Team Behind the Award
The ECR 2026 Best Research Presentation Abstract Award was received in Vienna by Martin Willemink, MD, PhD, our very own Co-founder & Chief Scientific Officer. Martin accepted the recognition at the Awards Ceremony in front of delegates from 121 countries.

The study was conducted by our multidisciplinary team across Segmed: Jie Wu, PhD (Co-founder and Chief of AI), Aline Lutz de Araujo, MD, PhD (Vice President of RWE and Clinical Insights), Martin Willemink and Sean Khozin, MD, MPH (Physician-scientist and CEO of the CEORT - CEO Roundtable on Cancer & Project Data Sphere), combining expertise in data science, clinical research, and radiology to design and execute the analysis at a scale that would not have been possible without Segmed's real-world imaging data platform.
The award, presented by the European Society of Radiology (ESR), recognizes the best-rated research presentation abstract per topic as selected by the ECR 2026 Scientific Subcommittees. Winners receive free registration to the subsequent congress alongside a certificate. But more importantly, recognition from one of the field's most respected bodies that the study is worth the community's attention.
That this honor was given to a study grounded in real-world data (not a controlled trial, not a simulation) is itself a signal. It reflects a growing recognition within radiology that understanding current practice is just as important as designing ideal practice.
What Is Oligometastatic Disease and Why Does Reporting It Matter?
Oligometastatic disease (OMD) describes an intermediate state of cancer spread: more than localized disease, but less than widespread metastasis. Typically defined as 1-5 metastatic lesions, OMD represents a clinically meaningful threshold because patients in this state may still be candidates for aggressive, curative-intent local therapies, including Stereotactic Body Radiation Therapy (SBRT), surgical metastatectomy, and thermal ablation.
The decision to pursue these interventions almost always begins with an imaging report. The radiologist's documentation of lesion count, location, disease trajectory, and prior treatment history directly shapes the multidisciplinary discussion that follows. When that documentation is consistent and precise, it opens doors for patients who might benefit from targeted treatment. When it is absent or imprecise, those opportunities may go unrecognized.

Radiology that consistently recognizes and communicates OMD - relevant features creates the conditions for optimal treatment selection. Radiology that does not may inadvertently close doors for patients who could benefit from curative-intent local therapy.
The Study: What Did 33.7 Million Reports Reveal?
The award-winning research conducted a large-scale retrospective analysis of real-world radiology reports drawn from across the United States, 33.7 million reports spanning 40 states. Researchers searched for mentions of oligometastatic disease using terminology consistent with the ESTRO-EORTC classification framework.
The results were revealing:
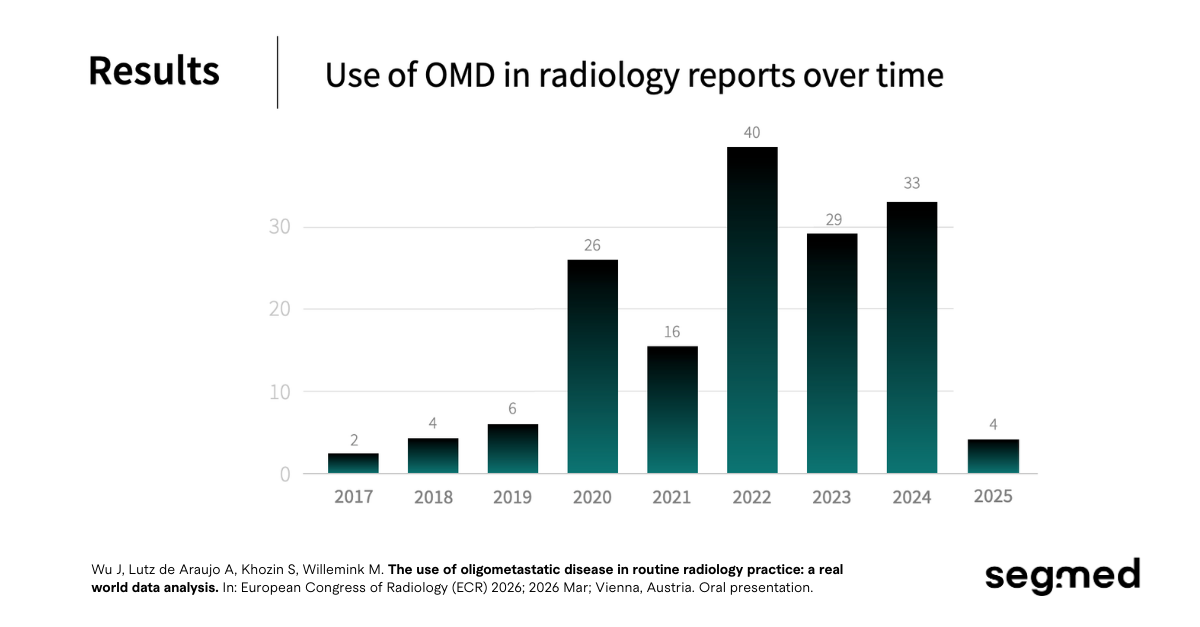
Out of the tens of millions of exams, only 164 reports were found that mentioned oligometastatic disease (OMD) or a variant (e.g. oligometastasis). Of the 164 reports that mentioned OMD, clinicians referring physicians, oncologists, and other non-radiology team members provided the OMD context in the indication or clinical history field in 148 reports from 95 patients. Radiologists independently used the term OMD in their own report narrative in just 18 reports from 17 patients.
That is the central finding of the study recognized with the Best Research Presentation Abstract Award at ECR 2026: a large-scale, real-world observational analysis of how oligometastatic disease (OMD) is used or more accurately, how rarely it is used in routine radiology practice across the United States.
Why Radiology Reports Are the Front Line for OMD
When a patient presents with limited metastatic disease, the decision of whether they might benefit from aggressive local treatment Stereotactic Body Radiation Therapy (SBRT), surgical metastatectomy, or thermal ablation typically begins with an imaging report. How that report frames lesion count, location, prior treatment history, and disease trajectory shapes the multidisciplinary discussion that follows. Radiology that consistently recognizes and communicates OMD-relevant features creates the conditions for optimal treatment selection. Radiology that does not may inadvertently close doors for patients who could benefit from curative-intent local therapy.
Why Real-World Data Is the Right Tool for This Question
Clinical trials, by design, select for ideal conditions. Patients are screened, definitions are standardized, and the clinical team is attuned to the specific criteria under study. Real-world data cuts through that filter. It reveals what happens when a concept moves from the protocol to the routine clinical practice.
For OMD, a real-world analysis of routine radiology practice offers something clinical trials cannot: an honest baseline. It revealed how often radiologists are using OMD-consistent language, which aspects of the ESTRO-EORTC classification framework are making it into reports, whether patterns differ by tumor type or imaging modality, and where education or structured reporting tools might produce the greatest gains.
This kind of work was singled out for recognition at ECR 2026 speaks to a broader disciplinary maturation. The field is not merely asking can radiology contribute to OMD management. It is beginning to ask, rigorously and empirically, how well is it currently doing so?
Recognition at the world's second-largest radiology congress sends a clear signal: translational, practice-anchored research is not a lesser endeavour: it is an urgent one.
The Role of Real-World Data Infrastructure
Studies like this one are not possible without the right data infrastructure. Analyzing 33.7 million radiology reports requires not only scale, but also the ability to search, filter, and surface signal across highly heterogeneous clinical documentation: different EHR systems, report formats, terminology conventions, and institutional practices.
This is precisely where platforms purpose-built for real-world medical data, like Segmed, become essential. Access to large, de-identified, multi-institutional datasets, along with the tools to query them effectively, is what transforms a clinically interesting hypothesis into a publishable, award-winning finding.
For research teams exploring questions in oncologic imaging, structured reporting, AI model development, or clinical decision support, the availability of this kind of data is not a background condition. It is the research.
What Comes Next: Structured Reporting and AI-Assisted OMD Identification
The findings from this study point directly toward two areas of opportunity for the radiology community:
1. Structured Reporting Templates
If radiologists are not independently documenting OMD-relevant features, one path forward is to make it easier for them to do so, through structured reporting templates that prompt for lesion count, prior treatment history, and disease trajectory in relevant oncologic contexts. The evidence base for designing these templates well now exists.
2. AI-Assisted Lesion Characterization
AI tools that automatically flag OMD-consistent findings, based on lesion number, location, and progression patterns, could provide real-time decision support at the point of interpretation. These tools, however, need to be grounded in what radiologists actually document, not what a trial protocol assumes they do. Studies like the one recognized at ECR 2026 provide exactly that foundation.
A Signal Worth Paying Attention To
The gap between how OMD is defined in clinical literature and how it appears, or doesn't, in routine radiology reports is not a minor documentation issue. It is a clinical gap with real consequences for patients who might otherwise be considered for curative-intent therapy.
The study recognized at ECR 2026 names that gap precisely, at scale, with real-world evidence. And in doing so, it sets the agenda for a new wave of research: investigating what it would take to close it.
For researchers, imaging informatics teams, and AI developers working in oncologic imaging, the question is now on the table. The data to begin answering it is available. What the field does next is what matters.
Note that some radiologists are using the concept of OMD without naming the term in their report. To improve the awareness of this concept, it is important that radiologists and clinicians adopt the term in their clinical work.
Work With Real-World Imaging Data
The research recognized at ECR 2026 was powered by access to millions of real-world radiology reports. If you're working on questions in oncologic imaging, structured reporting, or clinical AI, the data to do this kind of work exists.
Exploring Segmed's radiology dataset?
Our de-identified, multi-institutional imaging data spans 40+ U.S. states and more than 150 millions of imaging studies, ready for research, model training, and clinical insights.
Looking for a research partnership?
We work with academic medical centers, imaging AI companies, and life sciences teams to design and execute real-world evidence studies. If you have a clinical question, we can help you build the data foundation to answer it.
Want to see what's possible?
Request a demo and explore how Segmed's platform can support your next oncologic imaging project: from hypothesis to publication-ready findings.
→ Get in touch at segmed.ai.
Curious about the research? Please feel free to browse the ECR 2026 library of abstracts or find more information about the oral presentation session and abstract published here.
Study Citation:
Wu J, Lutz de Araujo A, Khozin S, Willemink M. The use of oligometastatic disease in routine radiology practice: a real world data analysis. In: European Congress of Radiology (ECR) 2026; 2026 Mar; Vienna, Austria. Oral presentation.
References
- Timur Izmailov, Sergey Ryzhkin, Gleb Borshchev, Sergei Boichuk. Oligometastatic Disease (OMD): The Classification and Practical Review of Prospective Trials. Cancers [Internet]. 2023 Oct 31;15(21):5234–4. Available from: https://doi.org/10.3390/cancers15215234
- Palma DA, Olson R, Harrow S, Correa RJM, Schneiders F, Haasbeek CJA, et al. Stereotactic ablative radiotherapy for the comprehensive treatment of 4–10 oligometastatic tumors (SABR-COMET-10): study protocol for a randomized phase III trial. BMC Cancer [Internet]. 2019 Aug 19 [cited 2020 Dec 6];19(1). Available from: https://doi.org/10.1186/s12885-019-5977-6
- Ost P, Reynders D, Decaestecker K, Fonteyne V, Lumen N, De Bruycker A, et al. Surveillance or Metastasis-Directed Therapy for Oligometastatic Prostate Cancer Recurrence: A Prospective, Randomized, Multicenter Phase II Trial. Journal of Clinical Oncology [Internet]. 2018 Feb 10;36(5):446–53. Available from: https://doi.org/10.1200/JCO.2017.75.4853
- Guckenberger M, Lievens Y, Bouma AB, Collette L, Dekker A, de Souza NM, et al. Characterisation and classification of oligometastatic disease: a European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. The Lancet Oncology [Internet]. 2020 Jan 1;21(1):e18–28. Available from: https://pubmed.ncbi.nlm.nih.gov/31908301/
- Richard, Lefkowitz RJ, Vaios Hatzoglou, Ma W, Juluru K, Mayerhoefer ME. Standardized Reporting of Oncologic Response: Making Every Report Count. Radiology [Internet]. 2022 Jun 1 [cited 2023 Apr 24];4(4). Available from: https://doi.org/10.1148/rycan.220042
- Navarro-Domenech I, Barry A, Tsai J, Ma G, Wong P. Challenges in the Understanding of Oligometastatic Disease in Clinical Practice. Cureus [Internet]. 2024 Oct 27 [cited 2024 Dec 19]; Available from: https://doi.org/10.7759/cureus.72500
- Lievens Y, Guckenberger M, Gomez D, Hoyer M, Iyengar P, Kindts I, et al. Defining oligometastatic disease from a radiation oncology perspective: An ESTRO-ASTRO consensus document. Radiotherapy and Oncology: Journal of the European Society for Therapeutic Radiology and Oncology [Internet]. 2020 Jul 1 [cited 2021 Sep 29];148:157–66. Available from: https://pubmed.ncbi.nlm.nih.gov/32388150/
- Szturz P, Vermorken JB. Steering decision making by terminology: oligometastatic versus argometastatic. British Journal of Cancer [Internet]. 2022 Jun 17 [cited 2026 Mar 16];127(4):587–91. Available from: https://www.nature.com/articles/s41416-022-01879-3
- Steenbruggen TG, Schaapveld M, Horlings HM, Sanders J, Hogewoning SJ, Lips EH, et al. Characterization of Oligometastatic Disease in a Real-World Nationwide Cohort of 3447 Patients With de Novo Metastatic Breast Cancer. JNCI Cancer Spectrum [Internet]. 2021 Jan 24 [cited 2025 May 21];5(3). Available from: https://doi.org/10.1093/jncics/pkab010
- Naik Vietti Violi, Rami Hajri, Haefliger L, Nicod-Lalonde M, Villard N, Clarisse Dromain. Imaging of Oligometastatic Disease. Cancers [Internet]. 2022 Mar 10 [cited 2024 Mar 7];14(6):1427–7. Available from: https://doi.org/10.3390/cancers14061427
- Szturz P, Vermorken JB. Oligometastatic Cancer: Key Concepts and Research Opportunities for 2021 and Beyond. Cancers [Internet]. 2021 May21 [cited 2021 Jun 6];13(11):2518. Available from: https://doi.org/10.3390/cancers13112518


